Expert Perspective: This pathological analysis is presented by Professor Seungho Baek. Prof. Baek examines the Pathophysiology of Cancer Cachexia and Mortality to clarify the systemic burden cancer imposes on the human body.
The Pathophysiology of Cancer Cachexia and Mortality involves both mechanical organ dysfunction and systemic metabolic failure. While cancer cells are not inherently toxic, their unregulated growth leads to the obstruction of vital systems (digestive, respiratory, circulatory). A primary driver of mortality is Cancer Cachexia, a multifactorial syndrome characterized by the involuntary loss of skeletal muscle and fat mass, driven by pro-inflammatory cytokines such as TNF-α and IL-6. This state increases the basal metabolic rate (BMR) and reduces treatment tolerance, accounting for nearly one-third of all cancer-related deaths.
A frequent question in clinical pathology is why a collection of a person’s own cells can lead to their demise. As Professor Seungho Baek, I posit that the Pathophysiology of Cancer Cachexia and Mortality is a dual process: a physical invasion of space and a chemical hijacking of the body’s energy reserves.
1. Beyond Toxicity: The Physical Disruption of Vital Organs
Cancer cells are not inherently “poisonous.” Instead, they kill through invasiveness and metastasis. As tumors grow, they physically occupy space reserved for healthy tissue, leading to mechanical failure of the body’s life-support systems:
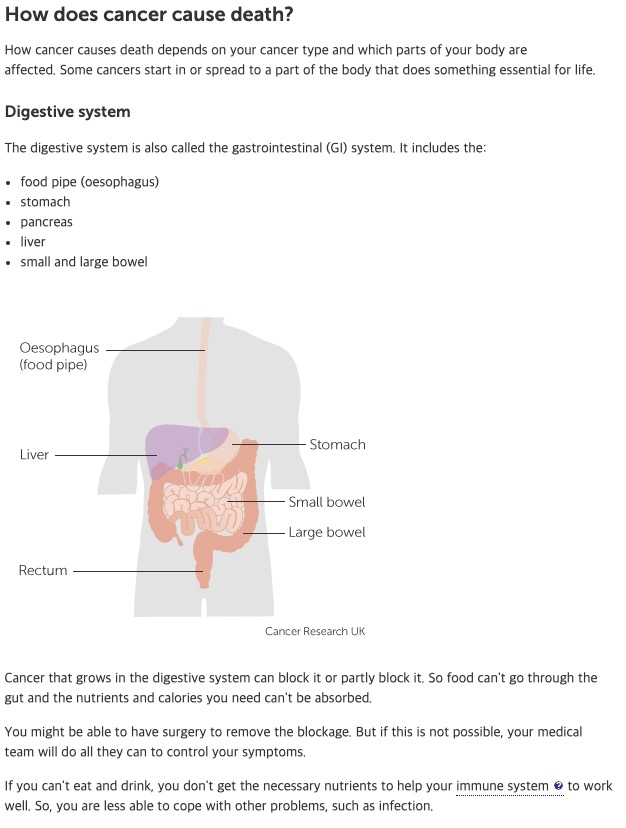
- Digestive Failure: Obstruction of the gastrointestinal tract leads to malabsorption and profound malnutrition.
- Respiratory Failure: Lung involvement impedes gas exchange and obstructs airways.
- Vascular and Liver Dysfunction: Tumors can invade blood vessels, causing clotting or internal hemorrhage, or impair the liver’s metabolic and detoxifying capacity.
2. Cancer Cachexia: The Wasting Syndrome
The most devastating aspect of the Pathophysiology of Cancer Cachexia and Mortality is the systemic wasting syndrome known as cachexia. Unlike simple starvation, cachexia cannot be reversed by increased caloric intake alone.
“Professor Seungho Baek Insight: In cancer cachexia, the body enters a hypermetabolic state. Pro-inflammatory cytokines produced by both the tumor and the host’s immune response—specifically TNF-α, IL-6, and PIF (Proteolysis-Inducing Factor)—trigger the breakdown of muscle and fat at an accelerated rate.”
This condition causes a paradox where the patient’s basal metabolic rate (BMR) increases even as their energy intake drops. This systemic exhaustion makes patients less able to tolerate chemotherapy, creating a vicious cycle of decline.
Metabolic and inflammatory markers of Cancer Cachexia.
3. The Epigenetic Context: Inflammation and Survival
Linking this to The Biology of Belief, we can see cachexia as the body’s ultimate “protection mode” gone wrong. When the internal environment is saturated with inflammatory signals, the cells shift from growth and repair to a primitive survival state. Understanding the Pathophysiology of Cancer Cachexia and Mortality requires addressing not just the tumor, but the systemic “belief” of the body that it is under a permanent state of emergency.
4. Current Management and Research Frontiers
While there is currently no absolute cure for cachexia other than complete tumor removal, Professor Seungho Baek highlights ongoing research in several key areas:
- Anti-inflammatory Interventions: Targeting specific cytokines to slow muscle wasting.
- Anabolic Hormone Therapy: Using agents to promote protein synthesis and maintain muscle mass.
- Personalized Nutrition: Specialized metabolic diets to support organ function under stress.
- Resistance Exercise: Utilizing physical therapy to maintain functional mobility and muscle integrity.
5. Conclusion: A Holistic View of Mortality
Understanding the Pathophysiology of Cancer Cachexia and Mortality helps clinicians and patients focus on quality of life. By managing the inflammatory response and physical obstructions, we can mitigate the systemic wasting that accounts for up to one-third of cancer deaths.
As Professor Seungho Baek, I urge a focus on early prevention and metabolic balance to ensure that the body’s internal environment remains a place of growth rather than a battlefield of inflammatory destruction.
